Introduction
Whether you are growing berries or other crops in soilless culture or even in soil, understanding the fundamental principles of plant nutrition and polyhouse management is essential. Key aspects such as water quality parameters, suitability of water for hydroponics, water quality standards, the role of various nutrient elements in crop physiology, nutrient mobility, and the impact of pH and EC on plant nutrition all play a critical role in crop performance.
A clear understanding of these basics helps growers make better decisions, avoid common mistakes, and achieve consistent crop growth and quality.
Critical Growth Factors for Berry Cultivation
In berry cultivation, the management of key factors such as water quality, light, VPD (Vapour Pressure Deficit), temperature, and nutrients plays a major role in overall crop performance. A deficiency or imbalance in even one of these factors can directly impact plant growth, development, and fruit quality.
Below is a detailed overview of the major factors that significantly influence the growth and development of berry crops.
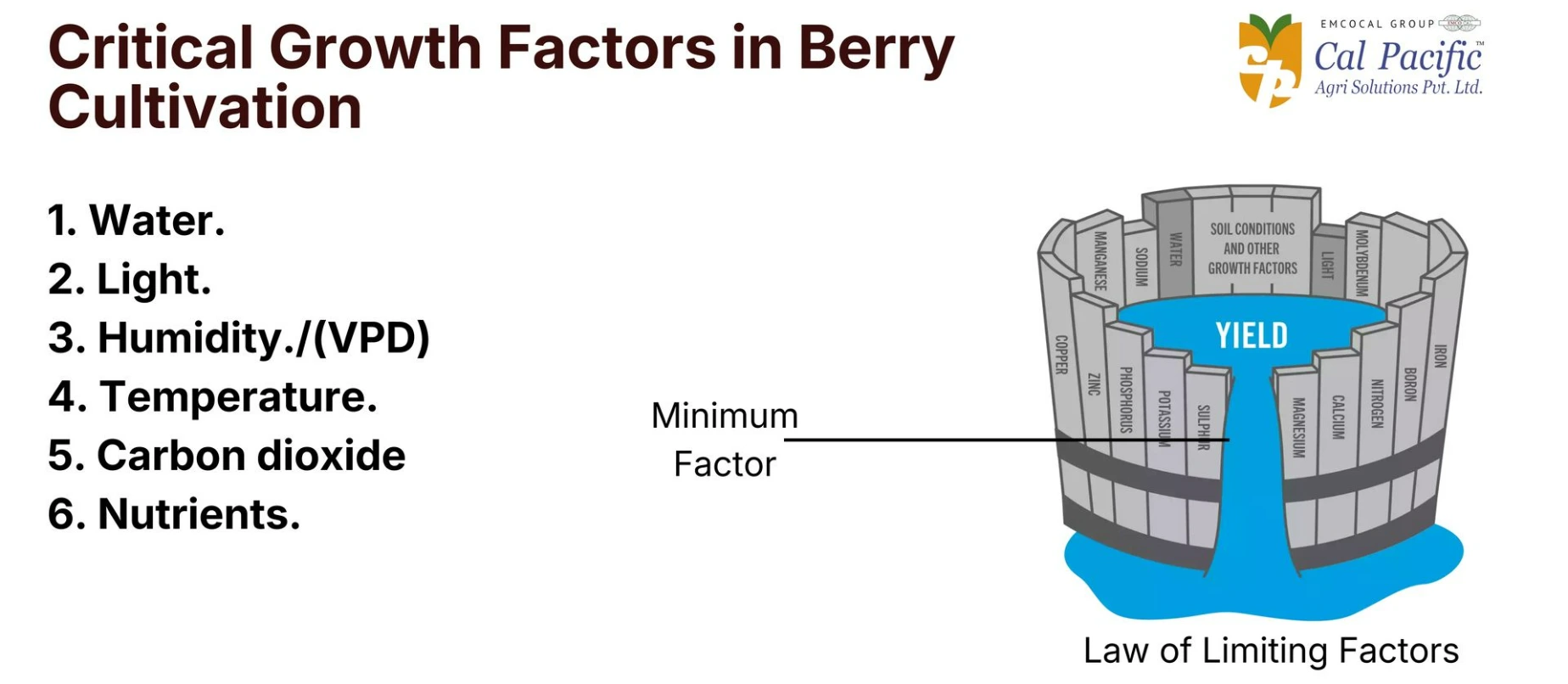
1. Water Quality Levels
The quality of irrigation water is a critical factor, especially in hydroponic systems, as it directly influences the composition and effectiveness of the nutrient solution. In soil-based cultivation, plants have some flexibility to access nutrients from the surrounding soil. However, in hydroponic systems, the root zone (rhizosphere) is confined and fully dependent on the supplied nutrient solution. This makes it essential to ensure that water quality remains within acceptable limits.
Rainwater is often considered ideal for hydroponic cultivation due to its low dissolved salts and natural purity. However, in practical conditions, irrigation water may contain various dissolved components such as sodium (Na) and chloride (Cl), which can affect plant growth if present in high concentrations.
Based on the levels of sodium and chloride, irrigation water can be classified into different quality levels, which helps in determining its suitability for berry cultivation and hydroponic use.
Quality Level | EC (mS/cm) | Na or Cl (mmol/l) | Na(ppm) | Cl (ppm) | Suitable uses |
1 | <0.5 | <1.5 | <34 | <53 | Suitable for all crops |
2 | 0.5-1.0 | 1.5-2.5 | 34-57 | 53-87 | Not suitable when recirculation is necessary |
3 | 1.0-1.5 | 2.5-4.0 | 57-92 | 87-142 | Not to be used for Salt-sensitive crops |
2.PH
The pH of the growing medium or nutrient solution plays a crucial role in nutrient availability and uptake. When the pH deviates from the optimal range, the uptake of certain nutrients becomes limited, even if they are present in the solution.
High pH levels can significantly reduce the availability of phosphorus and most micronutrients such as iron, zinc, and manganese, while molybdenum (Mo) remains relatively available. Continuous use of a high pH nutrient solution can lead to severe yellowing of plants, primarily due to micronutrient deficiencies caused by restricted availability.
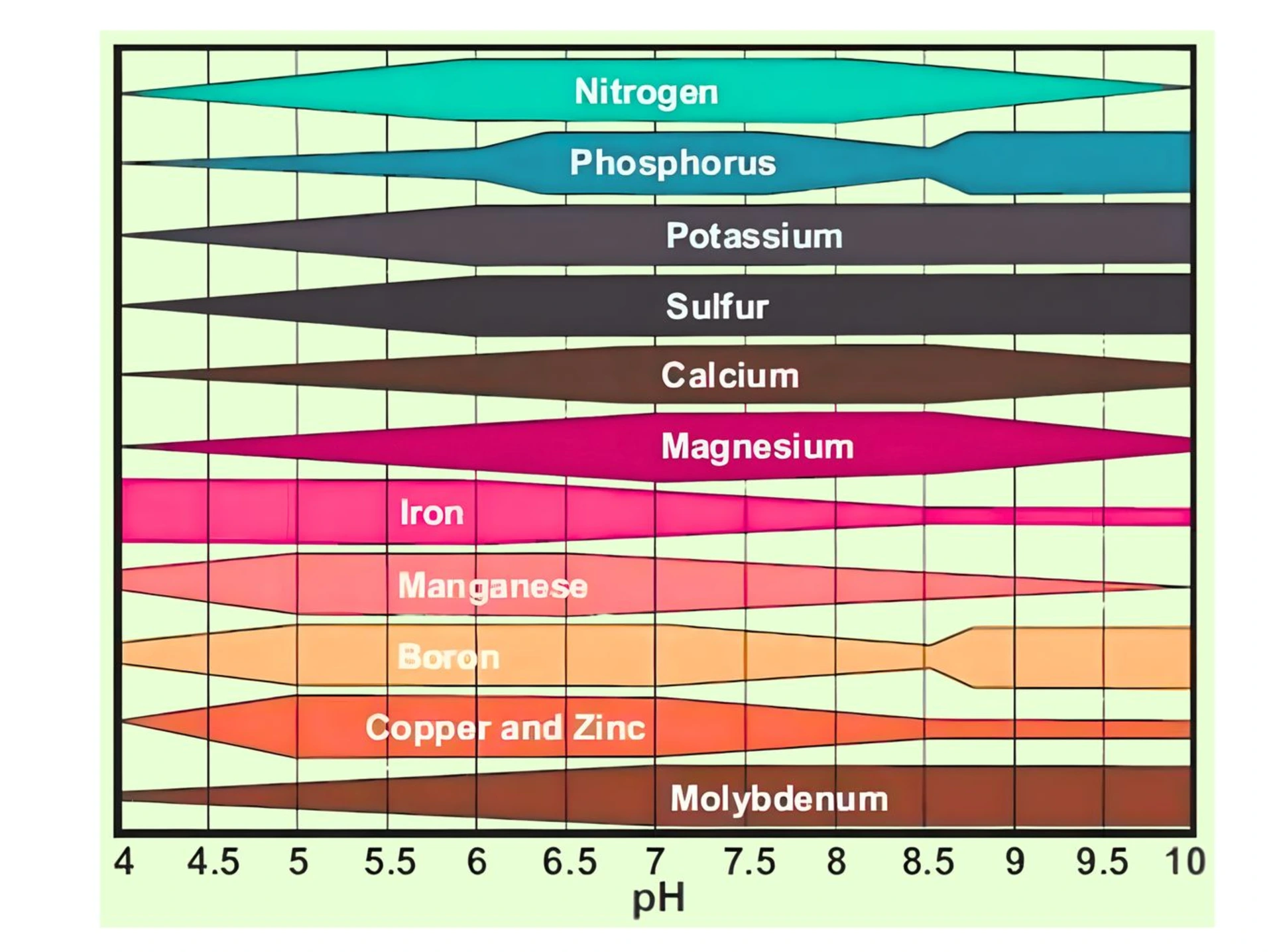
Managing High pH
To prevent and manage high pH levels in hydroponic systems, the following practices are recommended:
-
Regular Monitoring:
Frequently check the pH levels of the fertigation system and ensure that acid injection systems are functioning properly. Accurate dosing of acid is essential to maintain the desired pH range. -
Use of Ammoniacal Nitrogen:
The addition of ammoniacal fertilizers can help lower pH. However, this must be done carefully, as improper use can create further imbalance. In hydroponic systems, ammoniacal nitrogen should be maintained within 5–15% of the total nitrogen in the solution. Higher proportions can cause excessive pH drop, leading to additional nutrient issues. - Use of Acids for pH Correction:
- Acids such as nitric acid or phosphoric acid are commonly used to reduce the pH of irrigation water and nutrient solutions. However, acid application must be done carefully, with proper dilution and dosing, to avoid sudden pH fluctuations and potential damage to the crop or system.
3 .Electrical Conductivity (EC)
Understanding Electrical Conductivity (EC) is just as important as managing pH, as it directly reflects the concentration of nutrients in the solution. Proper EC management helps maintain a balanced nutrient environment for optimal plant growth.
EC plays a crucial role in the following:
-
Indicates Nutrient Concentration:
EC provides a direct measure of the total dissolved nutrients in the solution. -
Prevents Deficiency and Salt Stress:
Low EC can lead to nutrient deficiencies, while high EC can cause salt stress and reduced water uptake. -
Enables Crop-Specific Nutrient Control:
Different crops and growth stages require different EC levels, allowing for precise nutrient management. -
Helps Detect Salt Accumulation:
Monitoring EC in soil or substrate helps identify salt build-up over time. -
Supports Precise Fertigation Management:
EC acts as a key tool for adjusting nutrient supply based on plant requirements.
Regular monitoring of EC in both the feed solution and drainage (leachate) should be carried out on a daily basis. Based on these readings, timely adjustments and proper interpretation are essential to maintain optimal growing conditions.
4.Nutrient & Their Mobility:
All the macro and micro nutrients are very important for the normal plant growth in the absence of any one of the nutrient plant cannot maintain its normal growth and develops deficiency symptoms, affects metabolism and die prematurely. There are 16 essential plant nutrients such as C, H, O, N, P, K, Ca, Mg, S, Zn, Cu, Fe, Mn, B, Cl and Mo. In the absence of each of the essential elements, plants develop deficiency symptoms characteristic of the deficient element.
Based on the nutrient mobility in the phloem elements are classifies into three types
Sr . No | Mobility | Element |
1 | Mobile elements | N, K, P, S and Mg |
2 | Immobile elements | Ca, Fe and B |
3 | Intermediate | : Zn, Mn, Cu, Mo |
1. Nutrient Competition: Nutrient competition refers to the situation where two or more nutrients compete for the same uptake pathway in the plant roots. Since they use similar transport mechanisms, the presence of one nutrient in higher concentration can reduce the uptake of the other.
Example:
Potassium (K), Calcium (Ca), and Magnesium (Mg) often compete with each other for absorption.
2. Nutrient Antagonism :Nutrient antagonism occurs when an excess of one nutrient negatively affects the availability or uptake of another nutrient, leading to imbalance or deficiency symptoms even when nutrients are present in the solution.
Example:
Excess Potassium (K) can reduce the uptake of Calcium (Ca) and Magnesium (Mg).
3. Nutrient Synergism: Nutrient synergism is the positive interaction between nutrients, where the presence of one nutrient enhances the uptake or utilization of another.
Example:
Nitrogen (N) can improve the uptake of Magnesium (Mg), supporting better chlorophyll formation and plant growth.
An Excess of | May Cause a deficiency of |
Potassium | Calcium, Magnesium |
Sodium | Potassium, Calcium, Magnesium |
Calcium | Magnesium |
Magnesium | Calcium |
Iron | Manganese |
Manganese | Iron |
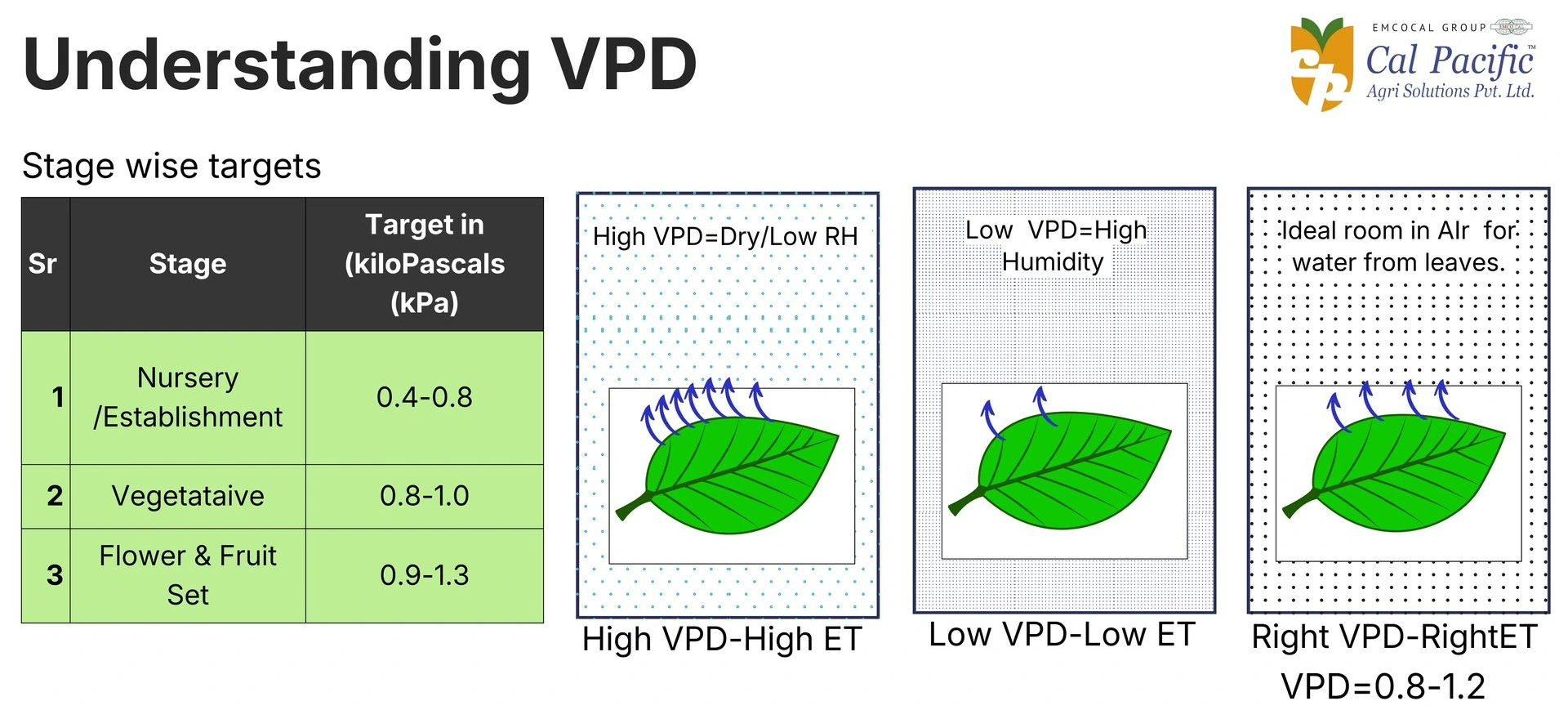
5.Vapour Pressure Deficit (VPD)
Vapour Pressure Deficit (VPD) is a key factor in polyhouse climate management that defines the difference between the moisture present in the air and the moisture the air can hold when saturated. It directly influences plant transpiration, nutrient uptake, and overall growth. When VPD is too low or too high, it can stress the plant and affect fruit quality. Maintaining an optimal VPD range ensures efficient water movement and balanced nutrient absorption in berry crops.
Conclusion
In berry farming, success depends on how well the basics are managed. Factors like water quality, pH, EC, VPD, and nutrient interactions are closely linked and directly impact crop performance.
Maintaining balance, consistency, and regular monitoring is key to achieving better yield and quality.